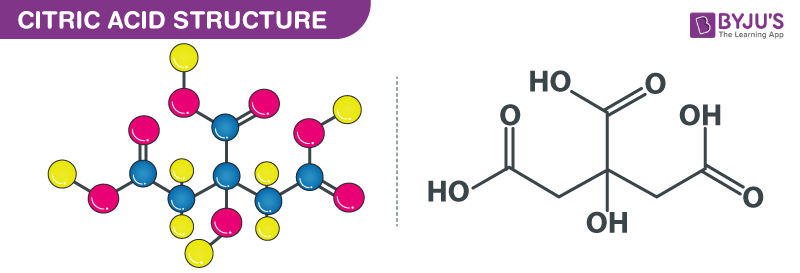
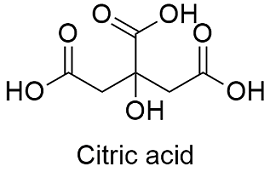
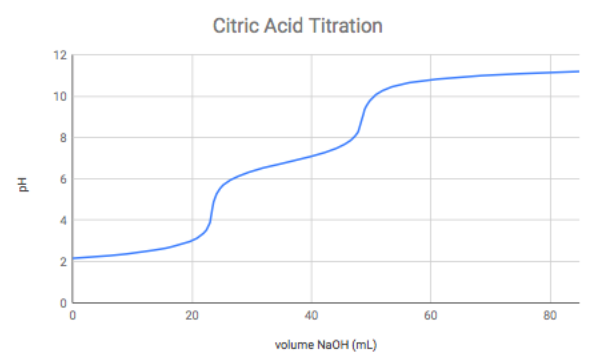
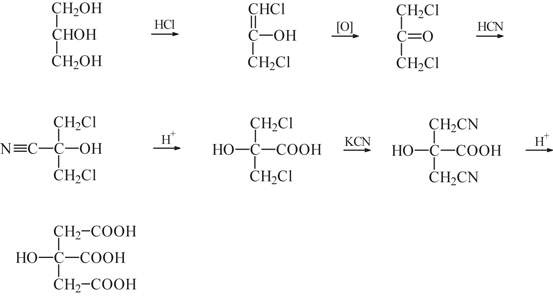
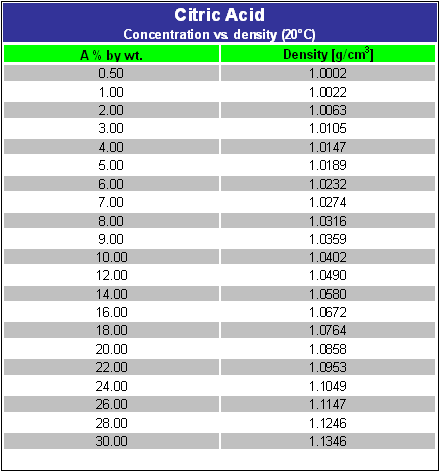
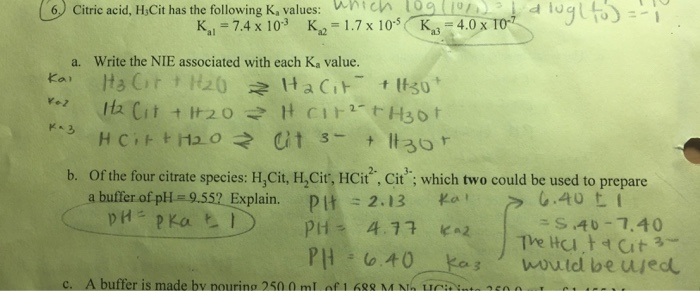Lemon juice normally has a `pH` of `2`. If all the acid the lemon juice is citric acid and there are - YouTube

SOLVED: Citric acid has 3 pKa values: 3.128, 4.761, 6.396. Calculate the Kf for the metal-EDTA complex: Number

Citric acid is a triprotic acid with Ka values of 3.14, 5.95, and 6.39. Estimate the pH of a 0.15 M solution of a monosodium salt. | Homework.Study.com
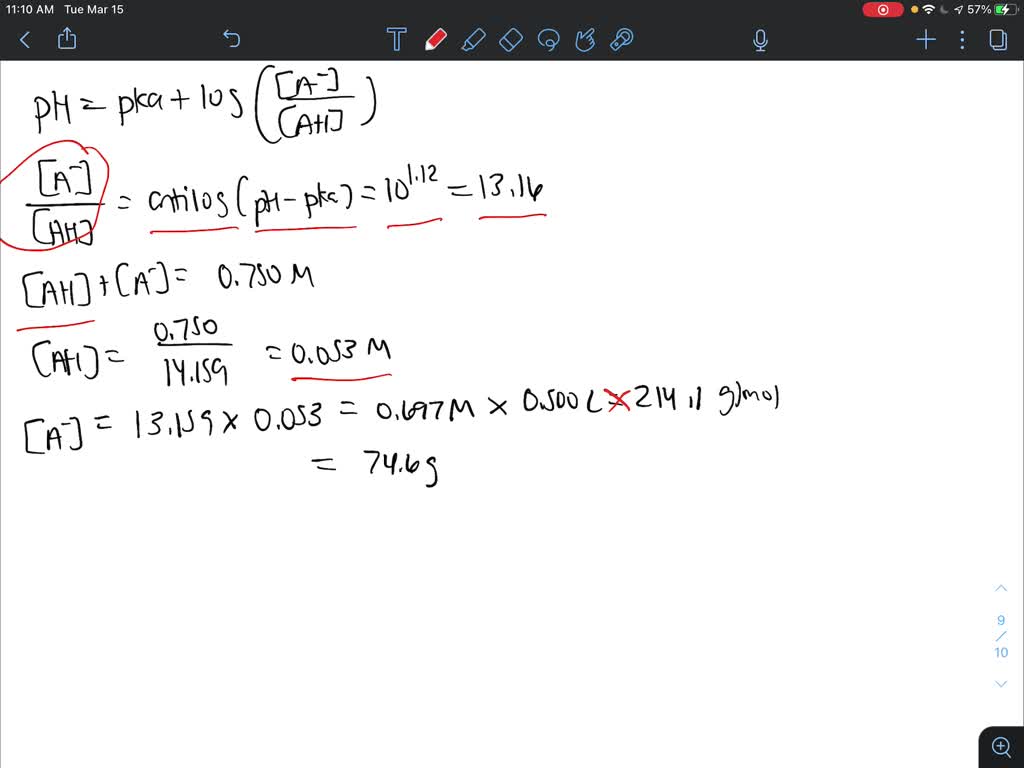
SOLVED: How will you prepare 500 ml of 0.750 M citrate buffer with pH of 4.25 from solid citric acid (C6H8O7) and solid sodium citrate (Na3C6H5O7).The Ka of citric acid is 7.40 x 10-4

SOLVED:Citric acid, which is present in citrus fruits, is a triprotic acid (Table 16.3) . (a) Calculate the pH of a 0.040M solution of citric acid. (b) Did you have to make