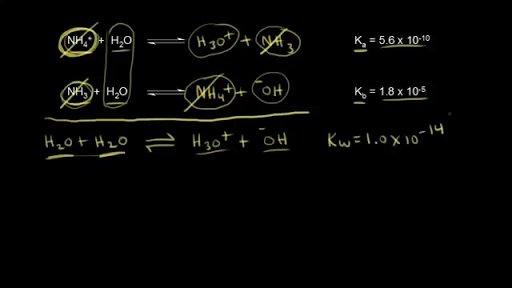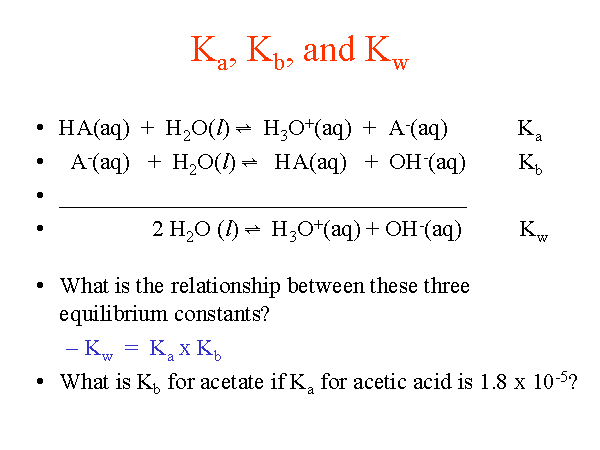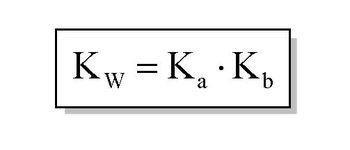From the dissociation constants Ka and Kb for an acid and its conjugate base, show that Ka· Kb = Kw .

How is Kw=Ka x Kb if each of those equilibrium constants include the concentration of another substance in them? Kw=H3o x oh but ka which is h3o also includes another substance? :

CALCULATING Ka Kb Kw, ICE Tables, ACID DISSOCIATION CONSTANT Power Points & M.C. WITH ANSWERS (59PGS) | Teaching Resources
What is value of Ka and kb for water (H20) and how numerically prove that ka ×kb=kw in case of water? - Quora

From the dissociation constants Ka and Kb for an acid and its conjugate base, show that Ka· Kb = Kw .
![SOLVED: Helpful Equations pH = -log [H2O+] Kw = Ka × Kb [ATJ[H2O+1 [HA]] = pOH -log [OH-] 14 = pH + pOH [HA][OH-] [A-] Kb Henderson-Hasselbalch Equation [A-] pH = pKa log - [HA] SOLVED: Helpful Equations pH = -log [H2O+] Kw = Ka × Kb [ATJ[H2O+1 [HA]] = pOH -log [OH-] 14 = pH + pOH [HA][OH-] [A-] Kb Henderson-Hasselbalch Equation [A-] pH = pKa log - [HA]](https://cdn.numerade.com/ask_images/9f4de4f19628424f8aea53cff3ecadcb.jpg)